Welcome to our Monthly Journal Club! Each month I post a paper or two that I have read and find interesting. I use this as a forum for open discussion about the paper in question. Anyone can participate in the journal club, and provide comments/critiques on the paper. This month’s paper is “Defined Paraventricular Hypothalamic Populations Exhibit Differential Responses to Food Contingent on Caloric State” by Michael Krashes and colleagues at The National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). I will provide a brief overview of the techniques/approaches used to make it more understandable to potential non-expert readers. If I am not familiar with something, I’ll simply say so.
Uncovering the neural populations responsible for appetite, feeding, metabolic control, and hedonic (rewarding/pleasurable) responses to food is essential for crafting new therapies for obesity, anorexia, and sickness-induced (e.g., cancer) appetite suppression. The hypothalamus is a critical structure controlling food intake and appetite, however the distinct roles of specific neural sub-populations in appetite control is not clear. If we can learn the ways different genetically-defined neural populations contribute to feeding, then we can potentially design drugs or other therapies that specifically target only those cells (to have the maximum effect with little off target problems).
The paraventricular hypothalamus (‘around the ventricle’; PVH) is an especially important hypothalamic node in appetite control, illustrated by the fact that lesions of this area cause massive obesity due to large increases in food intake. It is comprised of several neuromodulator populations (all expressing the critical transcription factor SIM1) that can be genetically targeted based on their expression of specific proteins: (1) glucagon-like peptide 1 receptor [Glp1r]; (2) melanocortin-4 receptor [Mc4r], (3) oxytocin [Oxt], and (4) corticotropin-releasing hormone [Crh]. These markers are not clear-cut, and many cells express more than one. Regardless, expression of these primary proteins demonstrates that each of these populations are largely independent from one another. The roles each of these play in appetite, food intake, and metabolism are unclear. Michael Krashes’ team tackled this problem using a variety of techniques including fiber photometry, immunohistochemistry, electrophysiology, and DREADDs.
The paraventricular hypothalamus (PVH; pictured above) contains many different unique cell types. The above image shows little overlap between two primary cell types in this region, those expressing melanocortin-4 receptor (Mc4r) and oxytocin (Oxt) (2.1% overlap). (Credit: Li et al., 2018)
They started by examining the activity of these genetically defined populations throughout the entire PVH following an overnight fast or after 2 hours of re-feeding (using the immediate early gene cFos). They observed state-dependent (fasted or re-fed) changes in cFos expression, with more cFos induction in the rostral (towards the front) portions of the PVN than the medial (middle) or caudal (towards the back) regions. Looking at specific subsets of cells expressing cFos following this experiment, they demonstrate that nearly all (except Oxt) neuronal populations examined show change in activity following fasting and re-feeding (see below). Notably, Glp1r-expressing neurons more than doubled their activity following re-feeding compared to the fasting state.
Induction of cFos in genetically-defined neuronal subtypes within the PVH after fasting or fasting followed by a 2-hour re-feeding session. As you can see, nearly all subtypes showed changes in activity (except Oxt neurons) with these manipulations. Some increased activity (Glp1r and Mc4r) while Crh neurons decreased their activity upon re-feeding. (cFos in RED with cell types in GREEN). (Credit: Li et al., 2018)
The researchers then attempted to delve deeper into this discovery through electrophysiological techniques. Using a very tiny electrode to monitor the activity of single cells in real-time, they were unable to find any differences in Glp1r neuronal electrical capacitance, resistance, holding current, or resting membrane potential. However, they did note that re-feeding increased the firing rate of these neurons, supporting their immunohistochemical (cFos) data. To move from single-cell to population level analyses, the researchers used adeno-associated viral vectors to express a fluorescent calcium indicator in their neurons of choice (using the Cre/Lox system). Then, they used fiber photometry to measure the activity of these cells in freely moving mice (see below). Calcium dynamics are proxy measures of neuronal activity, as calcium concentrations rapidly change when neurons are active.
Interestingly, the researchers found that Glp1r-expressing neurons were relatively silent during fasting, or when the mice were full. However, if food was presented to the mouse when it was hungry (fasted), these cells rapidly increased their activity! This suggests that the activity of these cells is not driven just by the availability of food, but the caloric/metabolic state of the animal (i.e., these cells act as a coincidence detector)!
Fiber photometry allows for direct imaging of neural population activity in freely moving mice. In panel (A) we can see the placement of the optical fiber in the PVH to record signals coming from Glp1r-expressing neurons. Panel (B) shows that these cells do not respond to new objects placed in the environment, but do respond strongly to food when the mouse is hungry (fasted). Notably these cells don’t respond to food when the mouse is full (fed). This effect is reduced when the food is inaccessible (minutes 0-20 of panel C), but pronounced when the mice can freely access the food (minutes 20-40 of panel C). In panel (D) we can see that this effect extends to high fat diet (HFD) in addition to the mouse’s regular chow. Crh-expressing neurons showed the opposite patten (i.e., their activity was suppressed upon chow presentation after fasting; not shown) (Credit: Li et al., 2018).
They repeated this experiment with all of the four cell types they examined previously. Mc4r- and Oxt-expressing neurons showed little change in these experiments. However, Crh-expressing neurons showed nearly the opposite pattern to that of Glp1r neurons, even though they are in the same brain area! This demonstrates that Glp1r and Crh neurons reciprocally track caloric state and respond in opposite patterns upon feeding after fasting. So what happens if the researchers manipulate these cells?
To do this, they used excitatory (Gq-coupled) or inhibitory (Gi-coupled or Kappa Opioid Receptor (KORD)) DREADDs. These designer receptors do not respond to any endogenous molecule in the brain, but do respond to the inert compound clozapine-N-oxide (CNO). This allows researchers to manipulate genetically-defined (e.g., Glp1r, Mc4r, etc..) neural populations with good temporal precision via systemic injections of CNO. They focused on Glp1r neurons as these showed the strongest responses during fasting and re-feeding. Activation of these neurons strongly suppressed appetite, while inhibition of these cells promoted feeding. Pre-treatment with the anorexic drug liraglutide (Lira) prevented this increased feeding response to inhibitory DREADD signaling, suggesting that Lira “can act redundantly at multiple sites and/or its action in the PVH is not critical in modulating food intake”.
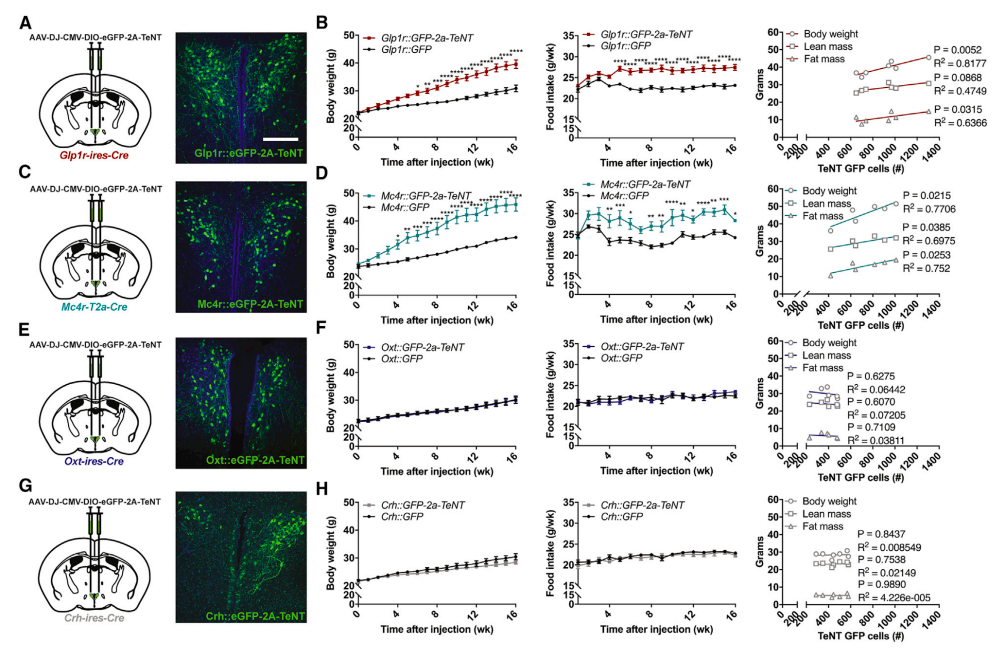
Finally, the researchers examined what happens when these different PVH populations are silenced over a long period of time (weeks and months), as all of their prior manipulations only looked at relatively short time-scales (minutes to hours). To silence these neurons, they used a virus encoding the tetanus toxin (see below), which permanently silences synapses primarily via cleavage of the protein synaptobrevin. They followed these mice for 16 weeks after viral injections, examining body weight and food intake throughout the course of the experiment (see below).
Tetanus toxin-induced chronic silencing of PVH neural subpopulations differentially affects body weight gain and food intake. In panels (A-B) we can see how silencing Glp1r neurons drastically increases body mass and food intake while panels (C-D) show a similar effect of Mc4r silencing. However, no effect was observed upon Oxt or Crh neuronal silencing. This suggests that PVH subpopulations do not serve redundant functions in weight gain and food intake. (Credit: Li et al., 2018).
Chronic silencing of Glp1r or Mc4r-expressing neurons strongly induced obesity and hyperphagia (overeating). These traits did not emerge in mice with silenced Oxt or Crh PVH neural populations. Further research needs to be done to understand how these diverse neural populations integrate and compute all that encompasses ‘appetite’ and ‘feeding’ (i.e., meal initiation, planning, satiation, and meal termination). This is a strong first step in understanding the differential responses and functions of these neurons, which will potentially lead to new treatments for metabolic diseases like obesity, anorexia, or sickness-induced appetite suppression. Future work should aim to map the afferent and efferent projections (incoming and outgoing connections) to and from these neurons, and find the critical downstream pathways controlling their anorexic or feeding effects.
Now join the discussion! Click the post title above and leave a comment!