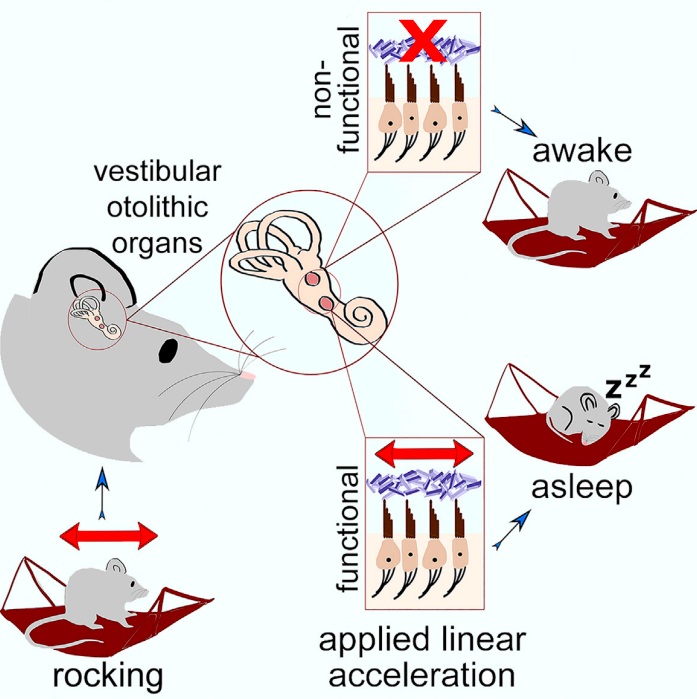
Welcome to our Journal Club! I use this as a forum for open discussion about the paper in question. Anyone can participate in the journal club, and provide comments/critiques on the paper by leaving a comment below. This is the 2nd post written totally by a guest author, lab member Emma Davidson! Her post discusses an interesting finding from a recent paper from Takashi Ogino and colleagues on molecular components of the circadian clock and breast cancer. Emma really took a deep dive into this paper and the surrounding literature, so this one is packed with lots of information. Check out the paper here!
Credit: Dana-Farber Cancer Institute
One of the great challenges of treating cancer is its resilience- aggressive cancers are hard to target and treat, and often recur despite our best treatments. The work of Ogino et al. further explores one of the hypotheses of why certain cancers are so resistant to treatments, and suggests a potential new target to create more effective cancer treatments against these aggressive and persistent variants. According to Tudoran et al., approximately 30% of early stage breast cancer cases relapse (2016). One of the reasons this might be is because of the heterogeneity of cancers, basically referring to the slight differences that occur in the genetic makeup between one cancer cell to the next. While this may seem trivial, these differences are often the result of various mutations that allow the cancers to evade different treatments, grow faster, or metastasize more. Therefore, the more heterogenous the cancer, the harder it is to treat because of the greater number of mutations it has accumulated.
There are 2 types of heterogeneity that are problematic in cancer treatment: tumor heterogeneity and intra-patient heterogeneity. Tumor heterogeneity refers to how different cells within the same tumor can acquire new mutations and carry slightly different genetic information than each other within the same tumor. Intra-patient heterogeneity is the differences in genetics that can occur in one tumor in the body compared to a different metastasis elsewhere in the body. Prior discussions of tumor and intra-patient heterogeneity of cancers have suggested 2 primary models to explain these observations: the non-hierarchal model, which suggests this arises because of clonal evolution, and the hierarchal model, which introduces the idea of “cancer stem-like cells”, or CSCs, in which some cancer cells have a multipotency that allows for them to act similarly to stem cells to continually drive tumor growth and allow for various types of cells to evolve (See Fig. 1)(Tudoran et al., 2016).
Figure 1: Schematic of non-hierarchal (A) vs hierarchal model (B) explanations of cancer heterogeneity. Note in the hierarchal model, “cancer stem cells” divide in a way that produces both new variants and self- renews the stem cell type. The non-hierarchal model accumulates mutations after divisions that lead to heterogeneity. (Tudoran et al., 2016)
With more investigation, Ogino et al. and colleagues have identified primitive stem cell-like cancer cells in breast cancers, which has led them to believe this may be the cause of heterogeneity in these kinds of breast cancer (2021). These breast cancer stem-like cells (BCSCs) exhibit various characteristics that help them resist “normal” treatments, such as remaining quiescent and in G0 phase for extended periods of time. Since G0 phase is a state in which cells are not actively dividing or preparing to divide, this behavior likely helps these cancer cells “hide” from the typical treatments that target rapidly dividing cells.
To make a bit of an oversimplification, cancer is essentially the unchecked division of cells; though there are many sneaky tricks that cancers accumulate to continue to grow and travel around the body. For this reason, the most typical kinds of cancer treatments target populations of cells that are rapidly dividing and proliferating. Since BCSCs do not act in this typical manner, they can escape these more typical treatments. BCSCs are then free to divide asymmetrically later with some daughter cells proliferating into the more typical tumor cells that rapidly divide, and others retaining this stem cell like character. This characteristic may be the reason that cancers recur- CSCs evade treatment when in their “dormant” (G0) state, making us think the cancer is all gone, but later give rise to the more typical cells that cause the tumor cells we are trying to fight off.
The current paper by Ogino et al. dives deeper into the problem of these CSCs in an attempt to better understand their underlying mechanisms and to create a new kind of treatment to target BCSCs specifically. Previous studies by the team investigated 4T1 cancer cells, a line of mouse breast cancer, and were able to group the cell line into 2 groups- those that behaved like “normal” tumor cells, and those that exhibited the above stem cell-like properties (a high capacity for self-renewal and differentiation) and were referred to as breast cancer stem-like cells, or BCSCs. These BCSCs identified within the 4T1 breast cancer line had abnormally high levels of aldehyde dehydrogenase (ALDH) activity and are considered ALDH+.
Upon further investigations, researchers have found such extensive links between stem-ness and elevated ALDH activity that high ALDH is now considered a marker for BCSCs. Notably, one commonly used breast cancer drug, cyclophosphamide, targets cells specifically with low ALDH activity, which may further exacerbate the problem of heterogeneity by allowing these stem-like cells to thrive. ALDH catalyzes the oxidation of aldehydes into carboxylic acids and is believed to help protect against oxidative stressors (such as reactive species like alcohols and aldehydes that our cells have to process and dispose of) as well as cytotoxic assaults elicited by chemotherapeutics (Edenberg & McClintick, 2018). High levels of this enzyme, ALDH, which catalyzes the process of detoxification therefore protects these cells from normal cancer treatments and promotes prolonged survival. Further observations by the research team revealed that ALDH activity varied in a time-dependent manner, which lead researchers to investigate possible molecular level interactions with the circadian system that might be correlated with upregulation of ALDH.
Figure 2. (Ogino et al., 2021) Graphical representation of relative amounts of ALDH+ cells within the 4T1 cancer cell line. The above shows that cells with increased expression of CLOCK protein, a key circadian regulator, had the lowest relative expression of ALDH activity. This indicates the probability of CLOCK having some inverse role in regulation or connection to ALDH expression levels.
To further examine the role that the circadian system might play in ALDH overexpression, authors inserted various key genes responsible for circadian rhythms into the 4T1 cancer cells and monitored the relative amounts of ALDH+ cells. They found that the cells with higher expression of the CLOCK protein had the greatest relative decrease in ALDH+ cells (Fig. 2), essentially showing that cancer cells with excess of this circadian gene do not have as elevated ALDH. Additional analysis of CLOCK mRNA and protein levels in ALDH+ vs. ALDH- cells revealed a significantly lower expression of CLOCK in the ALDH+ cells, indicating to researchers that the loss or down regulation of CLOCK may be implicated in the overexpression of ALDH and subsequent stem cell-like behaviors. A luciferase assay also confirmed lower CLOCK binding activity to its target genes, indicating less CLOCK activity in these ALDH+ cells.
So what if we “supplement” cancer cells with CLOCK?
The above preliminary investigations uncover an inverse relationship (that is, when one goes up, the other goes down) between CLOCK expression and ALDH+ cells with the associated cancer stem-like cell phenotype. The next step was then to see if this was a causal relationship or merely a correlation. To tell if the deficiency in CLOCK is the cause of ALDH overexpression and subsequent stem cell like properties, researchers devised a way to “supplement” the ALDH+ cells with the CLOCK gene to see if it would restore the “normal” cancer cell phenotype. Researchers found that using a specific virus to deliver the Clock gene into these ALDH+ cells was in fact a feasible way to increase functional CLOCK protein levels without disrupting the normal circadian fluctuations in its expression (Fig. 3).
Figure 3: (Ogino et al., 2021) Luciferase data showing the oscillatory circadian expression of CLOCK protein after transduction by the authors.
Figure 4. (Ogino et al., 2021) Western blot (top) and bar graph schematic of mRNA levels, showing much higher mRNA and protein expression in CLOCK transduced cells. p84 is a control used to normalize protein expression.
This finding was an important first step to confirm a way to supplement CLOCK to the ALDH+ cells without altering the way that CLOCK is normally expressed, which let authors further assess if supplying excess CLOCK was sufficient to restore a more typical cancerous phenotype (one that is treatable with current drugs) which would lower the overall fitness of the cancer. With this possibility, researchers began to see if supplementing CLOCK to these BCSCs could reduce the expression of ALDH, eliminate “stem-ness”, and subsequently mitigate the cancer’s ability to grow and spread. After lentiviral transduction (supplementation) of the CLOCK gene into these cancer cell lines, the authors found that the ratio of ALDH+/ALDH-negative cells was much lower, meaning there were far fewer cells that were overexpressing ALDH (Fig. 4). With this ability to manipulate the number of cells over expressing ALDH by supplementing CLOCK, researchers could begin to investigate if the overexpression of ALDH was indeed driving cells to act stem-like, or if this was just a correlation.
Figure 5 (Ogino et al., 2021) relative levels of mRNAs known to be associated with “stem-ness”. This figure shows how the cells supplemented with CLOCK have a much lower expression of “stem-ness factors”.
Upon analysis of mRNA expression within these cancer cell lines, populations with CLOCK expressed significantly lower amounts of “stemness” related factors, which are essentially markers for multipotency that offer a quantitative assessment of how “stem-like” a cell may behave (Fig. 5). Furthermore, these CLOCK transduced cells exhibited a slower growth rate and lower spheroid formation in culture, as shown in Fig. 6. Spheroids in this circumstance are populations of cells growing in 3D on media, which is a good testament to how well these cells would be able to grow in live tissue.
Figure 6: (Ogino et al., 2021) 2E- growth rates of cancer cells with CLOCK supplemented are significantly lower than those without supplemented CLOCK, 2F- Spheroid growth assay, showing the cell population with CLOCK supplemented have fewer spheroids growing (spheroids appear blue on images to the left), and smaller spheroids than the cell population without CLOCK supplemented (bar graph accounts for number and size of spheroids).
Keeping in mind the end goal, if we could target cancer treatments to attack the BSCSs, this could cause slower growth of the cancer over all by reducing the cancer’s ability to continually accumulate mutations that allow for faster growth and escape from common treatments, which could be an amazing way to treat heterogeneous cancers. But it is not just the growth rate of cancers that makes this disease so hard to treat, it is also the invasiveness of the cells (their ability to take over neighboring tissues), and the metastatic potential of the cells (their ability to migrate around the body, recolonize, and grow in new areas).
Upon assessment of the slower growth they were able to cause by supplementation of CLOCK (Fig. 6), researchers next turned their sights on the cancer’s invasiveness after CLOCK supplementation using two assays: a collagen type I TGF(beta)-1 induced invasion assay, and a spheroid invasion assay, both of which assess the cancer cell’s ability to grow and expand outward beyond where normal cells may stay put after being seeded (when cells are placed into the growing medium). After CLOCK transduction, both types of assays showed much less invasive potential (as seen in Fig. 7).
Figure 7: (Ogino et al., 2021) A. Invasion assay of cancer cells showing that populations receiving the CLOCK supplement are much less invasive(left images), with quantitative r=confirmation of less area being occupied by CLOCK supplemented cancer lines. B. Spheroid invasion assay images, showing qualitative observations of much less invasion and protrusion occurring from the cell spheres supplemented with CLOCK (bottom images). C. Western blot analysis of endothelial (less invasive cell type) markers E-cadherin and Claudin1, seen to be expressed more in CLOCK supplemented cells (right) than non-supplemented counterparts(left). Additionally, cancer cells not supplemented with CLOCK exhibit higher expression of mesenchymal marker, Vimentin (more invasive type).
Next, to assess the cancer’s migratory ability, researchers looked into markers that were indicative of whether the cells were in more of an endothelial or mesenchymal state. This is significant as EMT, or Endothelial to Mesenchymal Transition, which is a transition that cancer cells make to take on a more “migratory state”, further increasing invasive potential to reach distant sites of the body. Rather than being anchored to a certain area or tissue and exhibiting a more endothelial nature, after undergoing EMT cells begin to express mesenchymal factors rather than endothelial, which allows them to disconnect from whatever tissue they were connected to and travel in the bloodstream to begin colonizing other tissues. The finding that CLOCK-transduced ALDH+ cells expressed a significantly higher amount of endothelial markers rather than mesenchymal was therefore a good sign of reduced invasive potential, which was contrasted by the finding that populations not transduced with CLOCK expressed more mesenchymal markers (i.e., they were more invasive).
Seeing the success of CLOCK expression in vitro, experiments that use cells in a dish, researchers were able to begin testing his method in vivo by implanting these cancer lines into the mammary fat pads of mice to observe how this cancer would grow in a live animal after supplementing with CLOCK. The results found in vitro carried over into the in vivo model, with slower tumor growth, and less invasive potential. Taken together, these factors indicate an overall less malignant type of cancer when transduced with CLOCK in this cancer line (4T1). Figure 8 shows multiple assessments of tumor growth and malignancy, each of which supports the notion that CLOCK transduction leads to lower malignancy and growth rates. These measurements include measuring tumor diameter, immunohistochemistry (staining for a marker of cellular division), assessment of number and size of metastases in lungs, and colony growth after plating.
Figure 8: (Ogino et al., 2021)- A. Measurements and images of tumor volume after cancer cell implantation in mouse models. CLOCK supplemented cancer cell lines are consistently smaller than non-supplemented counterparts. B. Immunohistochemistry staining of CLOCK supplemented (bottom) vs. non-supplemented cancer cell lines. Authors stained for Ki-67+ cells (appearing pink/red), which indicates cells preparing for division and is frequently used to assess cancer progression. As seen qualitatively in images to the left, and quantitatively on the bar graph to the right, CLOCK supplemented cells have lower Ki-67+ levels, which is associated with a less severe cancer. C. Images and quantitative analysis of metastasis to the lungs after cancer cell injection shows CLOCK supplemented cancer cells exhibit less lung metastasis. D. Assessment of growth of metastatic colonies isolated from mice after cancer cell injection indicate less growth occurs with CLOCK induced cancer lines.
It works, but how?
After in vitro and in vivo experiments, authors could tell that CLOCK levels were much lower in these ALDH+ stem-like cancer cells, though supplementing CLOCK was sufficient to reduce stem cell like properties, leading to a slower growth rate and less metastasis. But how? To answer this, researchers examined whether CLOCK was repressed prior to, or after transcription, the process in which the information encoded by DNA is converted into mRNA and exported from the nucleus of the cell. There is only once copy of DNA per cell, so it must be “kept safe” and cannot leave the nucleus. Instead, cells have a system of copying the code of DNA into another similar molecule, RNA, that can leave the nucleus to transfer genetic information to the components of cells that will actually make the proteins encoded for in DNA. While it may seem redundant to make a copy of the genetic information in a slightly different molecule, it goes to show how important it is that DNA is kept safe in the nucleus and allows for just the needed region of DNA to be exported as often as needed. Though researchers knew that there was a lower amount of the CLOCK protein in these ALDH+ cells, they we not sure if it was because the DNA was not being encoded and exported to RNA (transcriptionally regulated), if the RNA was not getting translated into a protein (post transcriptional control) or, if the protein was being excessively degraded after its synthesis (post-translational).
To identify how CLOCK was being repressed in ALDH+ cells, researchers assessed levels of CLOCK mRNA in the ALDH+ cells compared to 4T1 cancer cells with normal ALDH expression. Given that the ALDH+ and ALDH- cells had similar levels of expression of the CLOCK mRNA, researchers could deduce that the regulation was not occurring transcriptionally, as both populations of the 4T1 cancer cells had similar levels of CLOCK mRNA. There had to be some mechanism in which the mRNA was inhibited from being translated into a fully functional CLOCK protein in the BCSCs expressing high levels of ALDH. As you might guess from the title, they found that CLOCK is repressed post transcriptionally in the ALDH+ cells through the binding of a micro RNA, miRNA-182. Essentially, this little non-coding RNA binds to the 3’ region of the CLOCK mRNA to prevent the mRNA from being translated into a functional protein. This shows how the problem is not in the CLOCK protein itself, but the regulatory mechanisms that impact its ability to act as a functional protein in the circadian system. Somehow, elevated ALDH levels are correlated with higher expression of this miRNA, which then binds to the CLOCK mRNA to inhibit its translation into a functional protein. See a brief overview of micro-RNAs below in Fig. 9.
Figure 9: Schematic of structure and function of miRNAs. miRNAs (orange) bind to mRNAs and inhibit their transcription by the ribosome, therefore stopping the associated protein from being expressed in the cell. Credit: Wikipedia; KelvinSong, Creative Commons.
Researchers were able to come to this conclusion by completing multiple luciferase assays, each targeting a different section of the regulatory regions of the CLOCK gene to determine where regulation of the CLOCK mRNA was occurring. By comparing the luciferase assays between ALDH+ and ALDH- cells, researchers could identify where regulation occurred by identifying the region that showed the biggest difference in luciferase expression between these two cell types. Upon identifying the location where regulation occurred, researchers were able to use this information and other known criteria of the mechanism of regulation to narrow down the known pool of miRNAs to identify which one may be imposing control over CLOCK expression. With the use of three specific criteria, Ogino et al. identified miRNA-182 as the miRNA responsible for post-translational inhibition of CLOCK in ALDH+ cells.
To confirm the role of miRNA-182 was indeed what they thought, researchers created a knock out (KO) cell line with depleted levels of miRNA-182, and injected these 4T1 cancer cells into mice. If miRNA-182 was responsible for the downregulation of CLOCK, depleting miRNA-182 should allow CLOCK levels to return to normal, and restore cancer cells to a less stem-like state. Compared to the unaltered 4T1 strain, mice that received an injection of miRNA-182 KO cells exhibited lesser tumor volume, fewer metastatic colonies, and a reduced area of metastatic colonies (as seen in Fig. 10) which supported the belief that miRNA-182 was responsible for downregulation of CLOCK. Additionally, expression of miRNA-182 was drastically elevated in tumor cells compared to other tissue types, which further supports the idea that this miRNA may play an important role in or be an effect of the elevation of ALDH, and subsequent stem-ness in this cancer cell population (Fig. 10).
Figure 10. (Ogino et al., 2021) Similar to Fig. 8 above, this figure assesses tumor size (A), metastasis (B), and metastasis growth (C), in each of which CLOCK supplemented cells grow slower and metastasize less. Panel D shows the abundance of miRNA-182 expression in tumor cells, further supporting the hypothesis that it may act in ALDH+ cancer cells to suppress CLOCK activity.
In total, the above work shows multiple ways that we may be able to target cancer stem-like cells of this particular cancer cell line. Authors show 2 main ways that reduced overall cancer growth: by supplementing CLOCK, or knocking out the miRNA that blocks CLOCK expression. While the expression of certain genes and miRNAs may not be the same in every cancer type, this paper shows an innovative way to re-examine cancer treatment by pinpointing a certain characteristic (in this case, “stem-ness”) and taking advantage of the complex interconnectedness of this characteristic with the molecular workings of circadian control.
Previous work has identified a somewhat ambiguous relationship between circadian control and cancer progression, though this current work takes it a step further by treating cancers indirectly through the ties and dysregulation that they cause to the molecular level circadian components. This “indirect” way of treating cancer may prove to be an incredibly valuable way to target specific attributes of different cancers based on each one’s individual properties and characteristics that aid in their malignancy and growth potential. In this case, attacking stem-ness through its connection with the circadian component CLOCK seems to mitigate growth rate and metastatic potential of the cancer by preventing cancer cells from accumulating mutations thanks to stem-cell like behavior (hierarchal model of heterogeneity), remaining dormant, and later reappearing and/or metastasizing. Though there is much that we still do not completely understand about cancers, and there are possibly other avenues separate from circadian components that could be leveraged to target idiosyncratic properties of other various cancers. See a summary figure of the authors’ findings in Figure 11.
Credit: Ogino et al., 2021
Annnnd that’s it for this post. Please make sure to leave a comment below and let us know what you thought of the paper and this write up. Until next time, stay curious!